
Maximise Treatment Outcomes. Minimise Treatment Barriers.
Provide your patients with an effective1 and easy-to-use, at-home depression treatment option.
Flow has a favourable side effect profile1,2 and can be used as a standalone intervention or in combination with other treatment approaches including medication.
Join more than 500 private practice clinics and 5 NHS trusts and build Flow into your depression treatment pathway.
Maximise Treatment Outcomes. Minimise Treatment Barriers.
Provide your patients with an effective1 and easy-to-use, at-home depression treatment option.
Flow has a favourable side effect profile1,2 and can be used as a standalone intervention or in combination with other treatment approaches including medication.
Join more than 500 private practice clinics and 5 NHS trusts and build Flow into your depression treatment pathway.


Treat Depression. Differently.
Flow is designed to optimise outcomes for both patients and clinicians by offering tailored solutions for each.
Patients are equipped with the Flow tDCS Headset and companion app, allowing them to conveniently manage their treatment from home. Clinicians benefit from a remote monitoring platform that provides real-time insights into patient adherence, outcomes, and supports data-driven care decisions.
Clinicians can adjust and customise patient stimulation schedules according to clinical need*.
*Subject to clinical qualifications and training
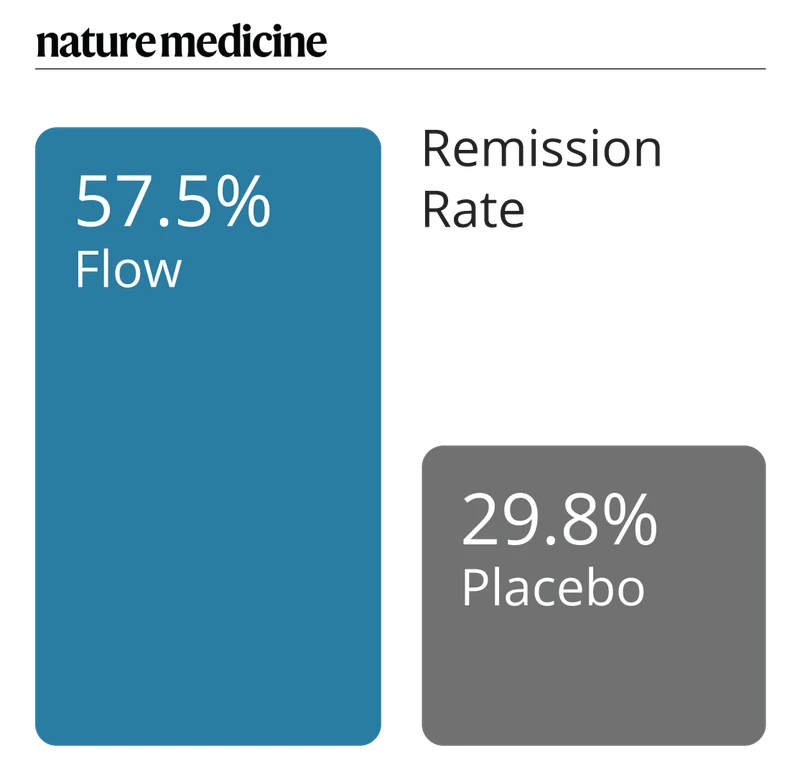
57.5% of study participants achieved remission in 10 weeks*
The recent EMPOWER clinical trial results, published in Nature Medicine, demonstrate Flow is effective and superior to the sham treatment1. This 10-week double-blind placebo-controlled randomised study reviewed the use of Flow compared to placebo across 173 adult participants diagnosed with unipolar depression (moderate to severe). Real-world data2 and various ongoing pilots3 show similar results.
The EMPOWER trial also highlighted a potential amplification when Flow was used in combination with an antidepressant. This impact has also been reported in real-world cases.
Could Flow be of interest to your patients looking for depression relief?
*Remission defined as a score of ≤12 on MADRS
Certified in the UK, EU, Switzerland, Norway, Hong Kong, Brazil and Australia.

Pilots in 5 NHS Trusts and NHS Practitioner Health

Approved by the British Standards Institutions (BSI)

Class IIa medical device CE-marked

Tested according to IEC 60601 for electrical medical device safety

Favourable Safety Profile
Depression treatment does not need to be a compromise. Longitudinal studies of tDCS report no major adverse events4. In the EMPOWER trial, there was increased reports in the active group of skin reddness, skin irritation, and trouble concentrating. Real-world evidence supports the favourable safety profile, with the most common side effects reported being skin reactions and headaches2.
Flow in practice


Over 30,000 real-world Flow users and 900,000 Flow stimulation sessions completed


300+ clinics offering Flow globally


Multiple NHS pilot programs from primary care settings to secondary care to NHS Practitioner Health
Who is right for Flow?
Flow is approved for use in anyone 18 or older with a Major Depressive Disorder diagnosis.
Flow has been provided to patients across Primary Care, Perinatal Services, Crisis Services and Community Mental Health settings.
What Clinicians say

Having used Flow with several patients over the past few months, I am very impressed with having this as an option to offer, with the responses I am seeing, and how patient-friendly the treatment/technology is. I think this is a real breakthrough moment in a speciality where so many of our medications can cause significant side effects, and therapy is not a "quick fix".
Dr. Hannah Nearney
Consultant Psychiatrist, Partner at Anchor Psychiatry Group

We are seeing rapid improvements in patients who have been struggling with severe depressive symptoms for many years, and it is allowing us to treat more patients at home in a far better environment to promote recovery in their mental health journey.
Dr. Mark McConnochie
Consultant Psychiatrist, Leicestershire NHS Partnership Trust Crisis Service

I use Flow with my clients who are stuck. Flow enables them to move forward and out of the loop of helplessness. They open up and see things they couldn't before. When they open up, they are more engaged in therapy.
David O’Farrell
Psychotherapist and Director at Mind & Body Solutions

Flow has received overwhelmingly positive feedback from our patients, who have reported improvements in their emotional well-being, increased energy levels, and overall functioning. Patients experience minimal discomfort or side effects during its use. Some patients have come off their antidepressants as a result of using Flow.
Dr. Azhar Zafar
General Practitioner, Northamptomshire Healthcare NHS Trust, Danes Camp Medical

The Flow application is easy to use as is the headset, the treatment can be conducted from home and patients find this a very useful treatment……The use of neuromodulation techniques such as tDCS and TMS provide an alternative and effective treatment for that group of patients who are very difficult to manage with medication.
Professor Alex O’Neill-Kerr
Consultant Psychiatrist, Clinical Director Transforming Mind Solutions and Northamptonshire Community Mental Health Team

Flow's exciting and encouraging data raise the prospect of a real advance in the treatment of depression...a helpful intervention for many people suffering from major depression.
Dr. Allan Young
Chair of Mood Disorders, King's College London

Having been used to offering rTMS to patients as a last resort, it is very liberating to be able to offer Flow as one of several initial treatment choices. Patients find it very easy to use and I love the fact that I can monitor their use using a validated scale Regarding the Flow RCT: These results are really exciting and support the clinical experience I have gained with Flow. Flow is hugely popular with patients and many are happy to use it when given the choice between it and another antidepressant.
Dr. Danny Allen
Consultant Psychiatrist, Clinical Director at Phoenix mental health services
See Flow in Action
Neuromodulation is an emerging field for many clinicians. To ensure you are fully supported, we provide complimentary training, ongoing clinical support, and a demo device, allowing you to evaluate Flow firsthand. At Flow, we are committed to broadening access to clinically effective mental health treatments, empowering clinicians to offer innovative care options for their patients.
Discover how Flow can integrate into your practice and enhance patient outcomes—request a demo today.

